 
Mendeleev put the elements in order of their relative atomic mass, and this gave him some problems. 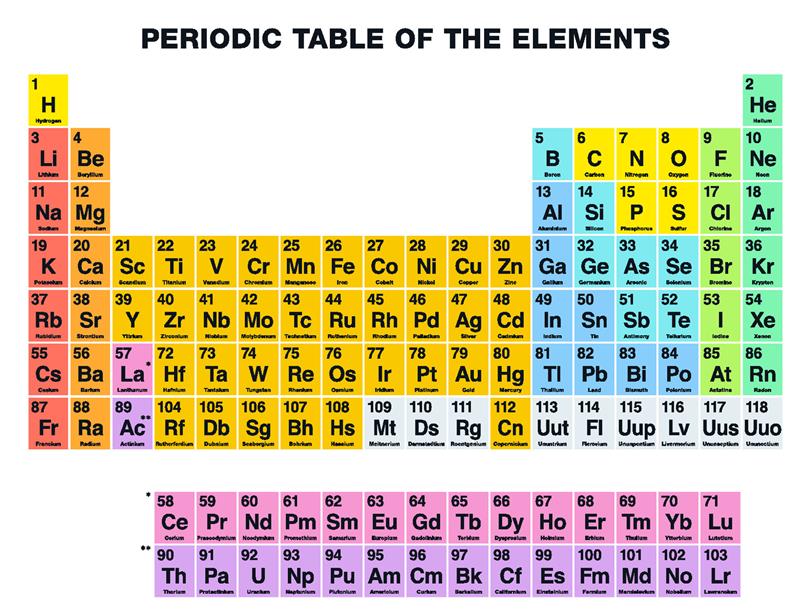
Mendeleev’s table needed one important modification before it became the modern periodic table – the use of atomic number to order the elements.Īll atoms of the same element contain the same number of particles called protons, and this is called the element’s atomic number. Two other predicted elements were later discovered, lending further credit to Mendeleev’s table. When this element, called gallium, was discovered in 1875, its properties were found to be close to Mendeleev’s predictions. For example, he predicted the properties of an undiscovered element that should fit below aluminium in his table. And when they were discovered, Mendeleev turned out to be right. He was also able to work out the atomic mass of the missing elements, and so predict their properties. But instead of seeing this as a problem, Mendeleev thought it simply meant that the elements which belonged in the gaps had not yet been discovered. Sometimes this method of arranging elements meant there were gaps in his horizontal rows or ‘periods’. Mendeleev also arranged the elements known at the time in order of relative atomic mass, but he did some other things that made his table much more successful. He realised that the physical and chemical properties of elements were related to their atomic mass in a ‘periodic’ way, and arranged them so that groups of elements with similar properties fell into vertical columns in his table. In 1869, just five years after John Newlands put forward his law of octaves, a Russian chemist called Dmitri Mendeleev published a periodic table. Find out how Mendeleev was able to predict elements that had not yet been discovered, and how the periodic table has accommodated dozens of new elements that have since been discovered. Dmitri Mendeleev organized the elements by families with similar properties, as well as by relative weight. In this excerpt from NOVA’s “Hunting the Elements,” see how the periodic table of elements took shape….
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
